How Do You Know When an Ion Hydrolyzes
Acid-Base Equilibria
86 Hydrolysis of Salts
Learning Objectives
By the end of this section, you will be able to:
- Predict whether a salt solution will exist acidic, basic, or neutral
- Calculate the concentrations of the various species in a salt solution
- Describe the acid ionization of hydrated metal ions
Salts with Acidic Ions
Salts are ionic compounds equanimous of cations and anions, either of which may be capable of undergoing an acid or base ionization reaction with water. Aqueous common salt solutions, therefore, may exist acidic, basic, or neutral, depending on the relative acid-base strengths of the salt's constituent ions. For example, dissolving ammonium chloride in h2o results in its dissociation, every bit described by the equation
![]()
The ammonium ion is the conjugate acid of the weak base of operations ammonia, NH3, and so it will undergo acid ionization (or acid hydrolysis):
![]()
Salts are ionic compounds composed of cations and anions, either of which may be capable of undergoing an acid or base ionization reaction with water. Aqueous table salt solutions, therefore, may exist acidic, basic, or neutral, depending on the relative acrid-base of operations strengths of the common salt's constituent ions. For example, dissolving the ammonium chloride in h2o results in its dissociation, equally described by the equation
![]()
The ammonium ion is the cohabit acid of the base ammonia, NH3; its acrid ionization (or acid hydrolysis) reaction is represented by
![]()
Since ammonia is a weak base of operations, K b is measurable and K a > 0 (ammonium ion is a weak acid).
The chloride ion is the conjugate base of hydrochloric acid, so its base ionization (or base hydrolysis) reaction is represented by
![]()
Since HCl is a strong acid, Chiliad a is immeasurably large and K b ≈ 0 (chloride ions don't undergo appreciable hydrolysis).
Thus, dissolving ammonium chloride in water yields a solution of weak acid cations (![]() ) and inert anions (Cl−), resulting in an acidic solution.
) and inert anions (Cl−), resulting in an acidic solution.
Computing the pH of an Acidic Table salt Solution Aniline is an amine that is used to manufacture dyes. It is isolated as anilinium chloride, ![]() a salt prepared by the reaction of the weak base aniline and hydrochloric acid. What is the pH of a 0.233 Grand solution of anilinium chloride
a salt prepared by the reaction of the weak base aniline and hydrochloric acid. What is the pH of a 0.233 Grand solution of anilinium chloride
![]()
SolutionThe K a for anilinium ion is derived from the Grand b for its conjugate base of operations, aniline (see Appendix H):
![]()
Using the provided information, an ICE table for this system is prepared:
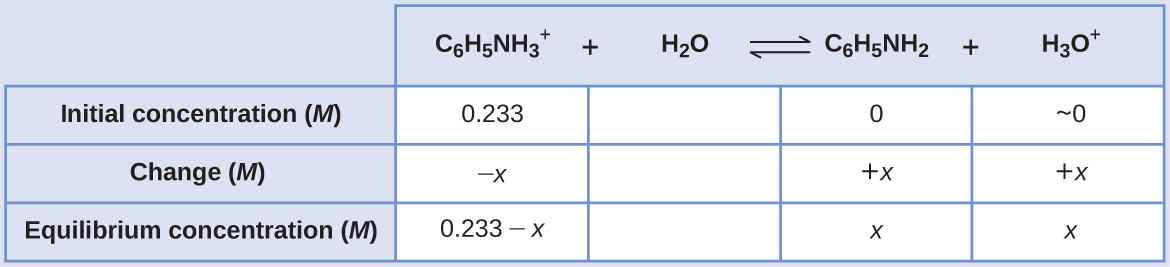
Substituting these equilibrium concentration terms into the K a expression gives
![]()
Assuming x << 0.233, the equation is simplified and solved for 10:
![]()
The ICE tabular array defines x every bit the hydronium ion molarity, and and then the pH is computed as
![]()
Check Your LearningWhat is the hydronium ion concentration in a 0.100-Chiliad solution of ammonium nitrate, NHivNO3, a salt equanimous of the ions ![]() and
and ![]() Which is the stronger acid
Which is the stronger acid ![]() or
or ![]()
Respond:
[HthreeO+] = 7.5 ![]() x−6 M;
x−6 M; ![]() is the stronger acid.
is the stronger acid.
Salts with Basic Ions
As another example, consider dissolving sodium acetate in h2o:
![]()
The sodium ion does not undergo appreciable acid or base ionization and has no effect on the solution pH. This may seem obvious from the ion's formula, which indicates no hydrogen or oxygen atoms, simply some dissolved metal ions function equally weak acids, as addressed afterward in this department.
The acetate ion, ![]() is the conjugate base of acetic acid, CH3CO2H, and then its base ionization (or base hydrolysis) reaction is represented by
is the conjugate base of acetic acid, CH3CO2H, and then its base ionization (or base hydrolysis) reaction is represented by
![]()
Because acerb acid is a weak acid, its K a is measurable and Chiliad b > 0 (acetate ion is a weak base).
Dissolving sodium acetate in water yields a solution of inert cations (Na+) and weak base of operations anions ![]() resulting in a basic solution.
resulting in a basic solution.
Equilibrium in a Solution of a Salt of a Weak Acid and a Potent Base Decide the acetic acid concentration in a solution with ![]() and [OH−] = two.5
and [OH−] = two.5 ![]() ten−six 1000 at equilibrium. The reaction is:
ten−six 1000 at equilibrium. The reaction is:
![]()
SolutionThe provided equilibrium concentrations and a value for the equilibrium constant will allow calculation of the missing equilibrium concentration. The procedure in question is the base ionization of acetate ion, for which
![]()
Substituting the bachelor values into the 1000 b expression gives
![]()
![]()
Solving the in a higher place equation for the acerb acid molarity yields [CH3COiiH] = i.i ![]() ten−v G.
ten−v G.
Cheque Your LearningWhat is the pH of a 0.083-1000 solution of NaCN?
Salts with Acidic and Bones Ions
Some salts are composed of both acidic and bones ions, and and so the pH of their solutions volition depend on the relative strengths of these two species. Besides, some salts contain a single ion that is amphiprotic, and so the relative strengths of this ion's acid and base of operations graphic symbol volition determine its effect on solution pH. For both types of salts, a comparison of the K a and K b values allows prediction of the solution'south acid-base of operations condition, equally illustrated in the following example exercise.
Determining the Acidic or Basic Nature of Salts Make up one's mind whether aqueous solutions of the post-obit salts are acidic, basic, or neutral:
(a) KBr
(b) NaHCO3
(c) Na2HPO4
(d) NHfourF
SolutionConsider each of the ions separately in terms of its effect on the pH of the solution, as shown here:
(a) The M+ cation is inert and volition not impact pH. The bromide ion is the conjugate base of operations of a strong acid, and so it is of negligible base force (no appreciable base ionization). The solution is neutral.
(b) The Na+ cation is inert and will not affect the pH of the solution; while the ![]() anion is amphiprotic. The One thousand a of
anion is amphiprotic. The One thousand a of ![]() is iv.7
is iv.7 ![]() 10−11,and its K b is
10−11,and its K b is ![]()
Since K b >> K a, the solution is basic.
(c) The Na+ cation is inert and will not affect the pH of the solution, while the ![]() anion is amphiprotic. The K a of
anion is amphiprotic. The K a of ![]() is iv.2
is iv.2 ![]() ten−13,
ten−13,
and its Thou b is ![]() Because K b >> K a, the solution is basic.
Because K b >> K a, the solution is basic.
(d) The ![]() ion is acidic (see to a higher place discussion) and the F− ion is basic (conjugate base of the weak acid HF). Comparing the two ionization constants: Chiliad a of
ion is acidic (see to a higher place discussion) and the F− ion is basic (conjugate base of the weak acid HF). Comparing the two ionization constants: Chiliad a of ![]() is 5.6
is 5.6 ![]() x−ten and the K b of F− is i.4
x−ten and the K b of F− is i.4 ![]() x−11, and then the solution is acidic, since G a > One thousand b.
x−11, and then the solution is acidic, since G a > One thousand b.
Check Your LearningDetermine whether aqueous solutions of the following salts are acidic, basic, or neutral:
(a) M2COiii
(b) CaCl2
(c) KHiiPOfour
(d) (NH4)2COiii
Respond:
(a) basic; (b) neutral; (c) acidic; (d) basic
The Ionization of Hydrated Metal Ions
Dissimilar the grouping 1 and 2 metal ions of the preceding examples (Na+, Catwo+, etc.), some metal ions function equally acids in aqueous solutions. These ions are not just loosely solvated by water molecules when dissolved, instead they are covalently bonded to a stock-still number of h2o molecules to yield a complex ion (see chapter on coordination chemical science). As an example, the dissolution of aluminum nitrate in water is typically represented as
![]()
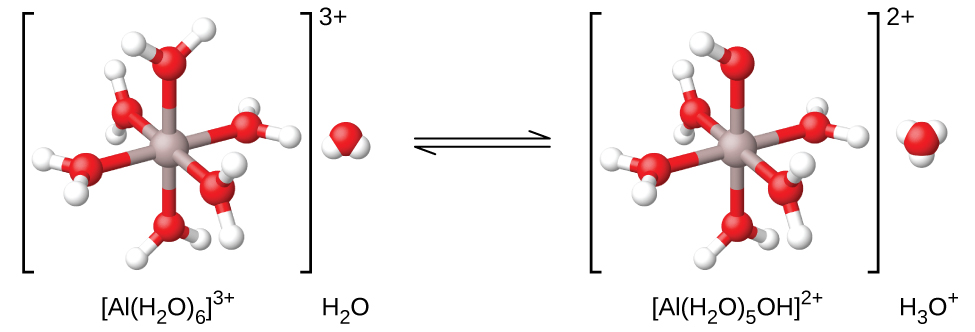
However, the aluminum(Iii) ion actually reacts with 6 water molecules to form a stable circuitous ion, and so the more than explicit representation of the dissolution process is
![]()
As shown in (Figure), the ![]() ions involve bonds between a key Al atom and the O atoms of the six water molecules. Consequently, the bonded water molecules' O–H bonds are more polar than in nonbonded h2o molecules, making the bonded molecules more decumbent to donation of a hydrogen ion:
ions involve bonds between a key Al atom and the O atoms of the six water molecules. Consequently, the bonded water molecules' O–H bonds are more polar than in nonbonded h2o molecules, making the bonded molecules more decumbent to donation of a hydrogen ion:
![]()
The conjugate base produced by this process contains 5 other bonded h2o molecules capable of interim equally acids, and so the sequential or step-wise transfer of protons is possible as depicted in few equations below:
![]()
![]()
![]()
This is an example of a polyprotic acid, the topic of discussion in a after section of this chapter.
When an aluminum ion reacts with h2o, the hydrated aluminum ion becomes a weak acid.
Bated from the brine metals (group ane) and some alkaline earth metals (grouping 2), nearly other metallic ions will undergo acrid ionization to some extent when dissolved in water. The acrid strength of these complex ions typically increases with increasing charge and decreasing size of the metal ions. The first-step acid ionization equations for a few other acidic metal ions are shown below:
![]()
![]()
![]()
Hydrolysis of [Al(HtwoO)vi]three+ Summate the pH of a 0.ten-Thousand solution of aluminum chloride, which dissolves completely to requite the hydrated aluminum ion ![]() in solution.
in solution.
Solution The equation for the reaction and K a are:
![]()
An Water ice table with the provided information is

Substituting the expressions for the equilibrium concentrations into the equation for the ionization constant yields:
![]()
![]()
Assuming x << 0.x and solving the simplified equation gives:
![]()
The ICE tabular array divers x every bit equal to the hydronium ion concentration, and so the pH is calculated to be
![]()
![]()
Check Your LearningWhat is ![]() in a 0.15-M solution of Al(NOthree)3 that contains enough of the strong acid HNO3 to bring [H3O+] to 0.ten Grand?
in a 0.15-M solution of Al(NOthree)3 that contains enough of the strong acid HNO3 to bring [H3O+] to 0.ten Grand?
Answer:
2.one ![]() x−v Thousand
x−v Thousand
Central Concepts and Summary
The ions composing salts may possess acidic or basic character, ionizing when dissolved in water to yield acidic or basic solutions. Acidic cations are typically the cohabit partners of weak bases, and basic anions are the conjugate partners of weak acids. Many metal ions bond to water molecules when dissolved to yield complex ions that may function every bit acids.
Chemistry End of Affiliate Exercises
Determine whether aqueous solutions of the post-obit salts are acidic, basic, or neutral:
(a) Al(NO3)iii
(b) RbI
(c) KHCO2
(d) CH3NH3Br
Determine whether aqueous solutions of the post-obit salts are acidic, basic, or neutral:
(a) FeCl3
(b) Thousand2CO3
(c) NHfourBr
(d) KClO4
(a) acidic; (b) basic; (c) acidic; (d) neutral
Novocaine, C13H21O2N2Cl, is the salt of the base procaine and muriatic acid. The ionization constant for procaine is 7 ![]() 10−6. Is a solution of novocaine acidic or basic? What are [H3O+], [OH−], and pH of a ii.0% solution by mass of novocaine, assuming that the density of the solution is one.0 g/mL.
10−6. Is a solution of novocaine acidic or basic? What are [H3O+], [OH−], and pH of a ii.0% solution by mass of novocaine, assuming that the density of the solution is one.0 g/mL.
Source: https://opentextbc.ca/chemistry2eopenstax/chapter/hydrolysis-of-salts/
0 Response to "How Do You Know When an Ion Hydrolyzes"
Post a Comment